Our Programs
HARNESSING THE IMMUNE SYSTEM
Through our pipeline of proprietary therapeutic candidates that target the immune response to cancer, we are developing the next wave of cancer medicines. Our programs span in vivo CAR, therapeutic mRNA, and antibody modalities.
TECHNOLOGY
ME Therapeutics has two myeloid cell targeted drug development programs and one drug discovery program currently underway. All three programs target distinct areas of myeloid cell biology in order to inhibit the suppressive effects of suppressive myeloid cells on the anti-cancer immune response. These drug candidates are being developed to target pathways of myeloid cell biology that are not currently being targeted effectively.
Drug Development
Our two development programs include our anti-G-CSF antibody candidate (h1B11-12) and our myeloid targeted prodrug candidates (D094 and D099).
Drug DISCOVERY
Our drug discovery program is focussed on the discovery of novel lipid nanoparticle formulations capable of effectively delivering small molecule drugs and or nucleic acids to myeloid cells in tumors. This program will support our existing prodrug development as well as potentially provide therapies for targeting myeloid cells in IO.
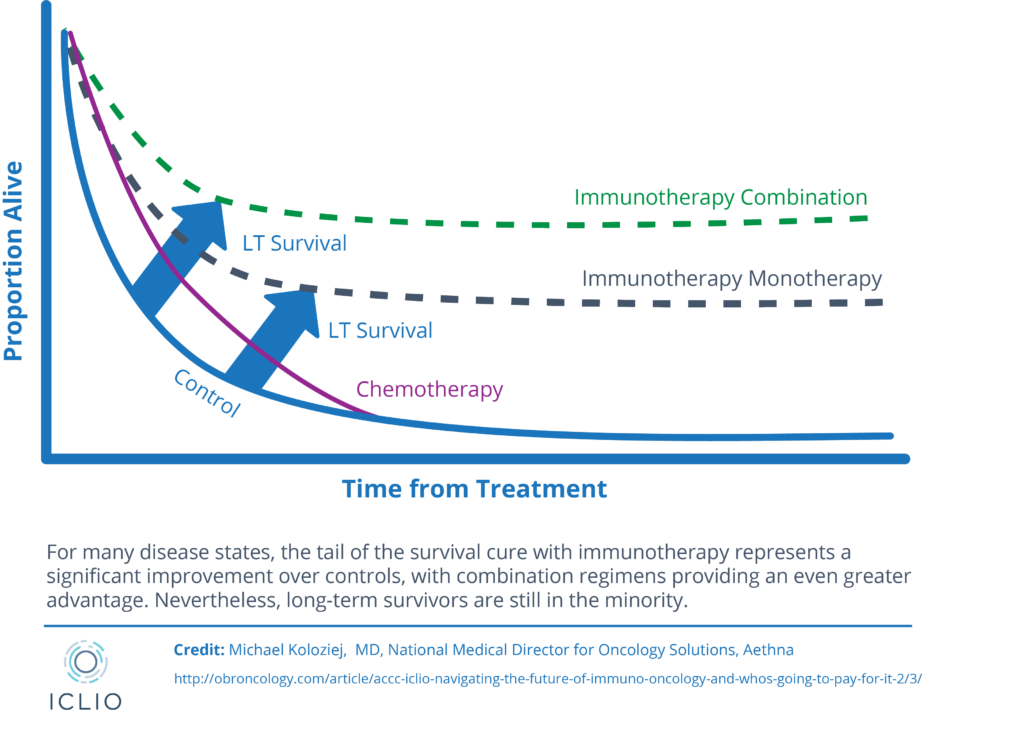
RESEARCH SHOWS
Combination Therapies Can Improve Patient Outcome. Current IO drugs (especially checkpoint inhibitors) have shown remarkable efficacy in some patients:
- Most cancer types are believed to be amenable to IO
- IO targets the immune system and not cancer directly
- Not all patients respond to single-agent IO
- Large market (estimated $154B by 2030)*
- Combining checkpoint inhibitors with myeloid cell targeting therapy has potential increase response rate
- Allow more patients to benefit from IO
- Target large unmet need
- Increase market share for existing therapies through more effective combinations
* Spherical Insights
The promise for immunotherapy oncology
IN VIVO CAR
We are discovering in vivo CAR therapies that deliver genetic instructions directly into the body so a patient’s immune cells can be reprogrammed to respond to cancer.
Our in vivo CAR program builds on the foundational success of standard CAR treatments and advances it further—equipping immune cells with a chimeric antigen receptor within the body rather than in a laboratory. This next-generation approach overcomes issues related to the toxic, expensive and cumbersome process of standard CAR therapy.
Our pipeline includes an in vivo CAR targeting CD22 for certain blood cancers, a CAR targeting a validated protein expressed in the tumor microenvironment in most solid tumors. Underpinning our program is a proprietary CD22 nanobody binder currently being evaluated in a Phase 1 study. We are also advancing a potentially universal CAR that may be used to target any cancer protein with existing antibodies. Underpinning
We have optimized our CAR constructs to be functional in both myeloid cells and T cells to potentially enhance efficacy in solid tumors by reprogramming both cell types. Testing has shown that our CARs function effectively for myeloid cell based killing of tumor cells and that the same CARs can lead to T cell activation.
THERAPEUTIC MRNA
We are developing therapeutic mRNA to help the immune system recognize and respond to cancer.
Our therapeutic mRNA program delivers instructions to cells in the tumour microenvironment to make proteins that attract immune cells to the area. The result is that “cold” tumours are turned “hot”. This means the body’s immune response to the tumours is activated.
Our proprietary mRNA therapeutic candidates target key, scientifically validated pathways—including the STING pathway and cytokine-driven pathways.
In a preclinical colorectal cancer model, our STING activator leads to the specific recruitment of immune cells into the tumor and significantly reduces tumor growth. Importantly, our data suggests that a single dose of our STING activator can synergize with a PD-1 checkpoint inhibitor, where the PD-1 inhibitor is ineffective on its own. This could mean that providing our STING activator may unlock the efficacy of checkpoint inhibitors in otherwise unresponsive tumors.
In addition, we believe this candidate may solve some of the past roadblocks observed with drugs targeting STING through the targeted delivery into myeloid cells in the tumor microenvironment, which could reduce systemic side effects.
G-CSF ANTIBODY PROGRAM
We are advancing a G-CSF antibody program that targets and blocks G-CSF, a cytokine protein known to drive immune suppression in the tumour microenvironment.
Our lead candidate, called h1B11-12, is a humanized antibody engineered to act on G-CSF. This protein is low or undetectable in healthy individuals but is transiently induced during acute inflammation. Tumours that make high levels of G-CSF create more immune-suppressing myeloid-derived suppressor cells (MDSCs) that prevent other immune cells from fully developing, which weakens the body’s ability to combat cancer.
Blocking G-CSF reduces these MDSCs, increases T cells and overcomes resistance to VEGF therapies. Together, this creates a more favourable immune environment to make tumours responsive to cancer treatment.
OUR INTELLECTUAL PROPERTY
PCT filed February 2018 on composition and use of lead anti-G-CSF antibodies (PCT/CA2018/050143) – Chinese patent granted in 2023.
Patents on prodrugs are expected to be filed once testing is complete – Initial search suggests freedom to operate based on proposed drug structures.
